Psychedelic Hype Cycle; Employer KAP Coverage; MDMA Cost-Evaluation Analysis; Onboarding Underrepresented Groups
This week we’re looking at a few developments that occupy what we might call the “infrastructure and operational” layer required for psychedelic-assisted therapies, including:
Employee sponsored coverage of ketamine assisted therapy
Cost Evaluation Analysis of MDMA for PTSD based on data from MAPS phase III
A novel approach to onboarding underrepresented groups into the psychedelic industry
Before we touch on these developments, I want to take a stab at placing this moment in the emerging psychedelic industry in a larger context.
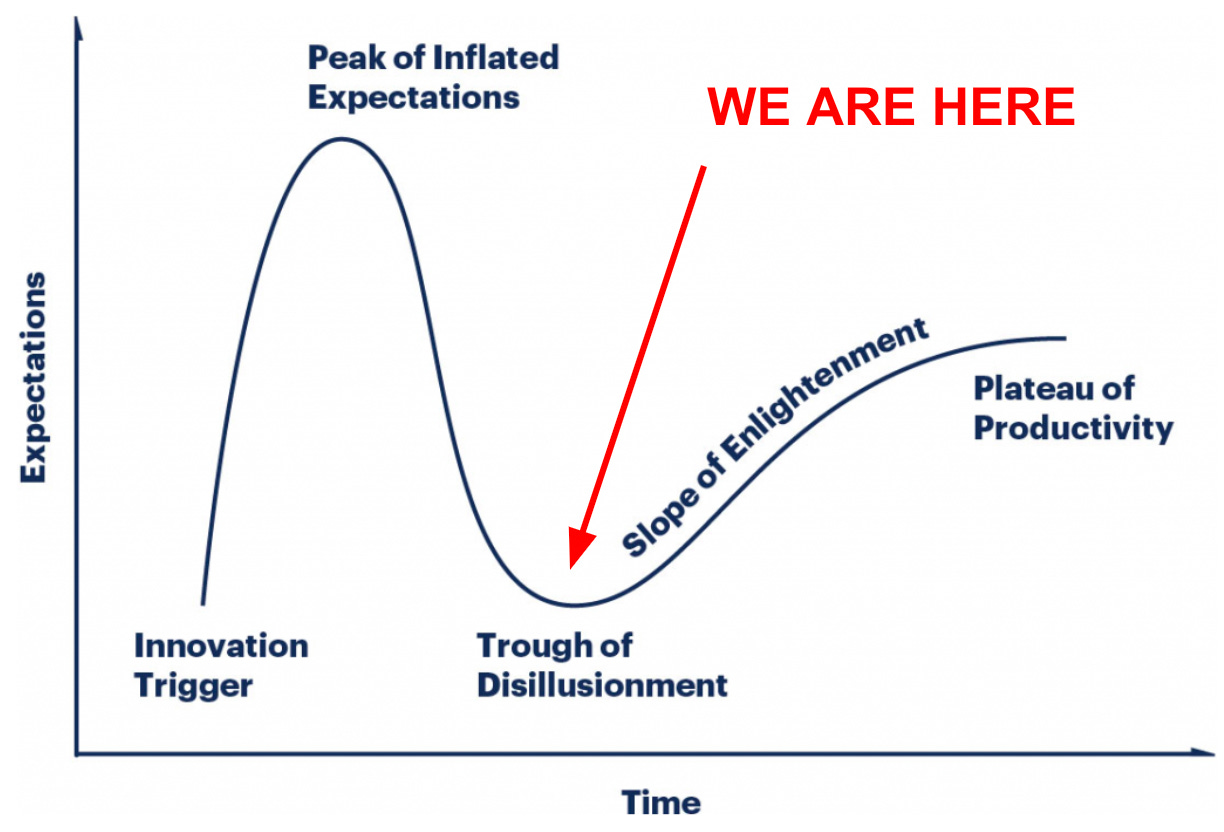
The Gartner Hype Cycle, which describes the narratives, waves of investment, and eventual adoption of new technologies, is a practical, even if cliched, framework for evaluating where we are in this collective psychedelic journey.
Last week, Dr. David Yaden employed this framework during talks at Imperial College London and Oxford University to describe the public perception of Psychedelics. Here I want to invoke the same framework to explore the narrative, vibe, and changing perspective of those working in the emerging industry day in and day out, rather than the public’s perception.1
Incubation Period: August 2017-December 2019
Unlike classical technological innovations like the automobile or the internet that have clearly defined Innovation Triggers, the psychedelic revival/renaissance lacks such a singular event.
However, nine seminal events between August 2017 and December 2019 validated the commercial, political, and cultural potential and led to the subsequent rapid explosion of interest, investment, and attention.
In chronological order:
August 2017—MAPS’ Breakthrough Therapy Designation for MAT for PTSD
May 2018—Publication of Michael Pollan’s How to Change Your Mind
October 2018—Compass Pathway’s Breakthrough Therapy Designation for Psilocybin for TRD
March 2019—FDA Approval of Spravato (esketamine)
April 2019—First University Psychedelic Research Center at Imperial College London
May 2019—Denver’s Decriminalization of Psychedelic Mushrooms by Ballot Initiative
June 2019 —Oakland Decriminalizes Psychedelics by City Council Resolution
September 2019— Launch of John’s Hopkins Psychedelic and Consciousness Research Center
November 2019—Usona Institute’s Breakthrough Therapy Designation for Psilocybin for MDD
These events confirmed for every broadly defined psychedelic subcommunity—investors, drug developers, philanthropists, scientists, activists, organizers—that psychedelics were going to be a thing.
The two years following this incubation period would see:
More than $2 billion invested
Almost sixty companies go public in Canada and America
Dozens of states and cities would propose, and many would pass decriminalization or legalization measures—including Oregon’s Measure 109
The launch of several psychedelic research centers at prestigious academic centers
Psychedelics would regularly appear in mainstream outlets like The New York Times, Business Insider, and even Town and Country Magazine.
The narrative driving the run-up to the Peak of Inflated Expectations is “psychedelics are a miracle cure” which is understandable given the:
Profound personal experiences these compounds potentiate
Promising results from early trials
A longstanding history of use throughout human history
Apparent absence of toxicity threshold and addiction potential
Associated stigma
Rising rates of depression and anxiety
Lack of mental health treatment options
However, after about two years of scaling the heights of hype, we are now squarely in the Trough of Disillusionment.
Evidence for this comes not only from bottomed-out psychedelic stocks but, more importantly, the recognition of a long list of challenges. Chief among them, and most jarring, are the instances of sexual transgression in psychedelic therapy.
Season 2 of Power Trip, by Psymposia’s Lily Kay Ross and Dave Nickles, launched this week with a harrowing account of sexual abuse of a participant in MAPS’ Phase II MDMA for PTSD trial. This same story was also the subject of a 2020 Quartz piece.
The field will need to grapple with this history and implement ethical standards, mitigation strategies, and transparent adjudication processes when these situations arise.
Also, as we noted in Reality Check: Reflections on Compass Pathways' Phase IIb Topline Results— what do we mean when we say psychedelics are safe?
“A vital feature of the Psychedelics as Medicine thesis is the safety narrative, precisely, the favorable toxicity profile and the characterization of being non-addictive—therefore, how can safety concerns peg back otherwise excellent results?
This depends on what do you mean by ‘safe.’
Even though psychedelics are currently considered non-toxic and non-addictive, the safety risk comes in the form of what might be called existential destabilization and hopelessness.”
Moving out of this trough will demand that the field better understand and respond to a broad array of challenges, including:
Psychological risks
Physiological risks such as Hallucinogen persisting perception disorder (HPPD)
The practical challenges of implementing legalization frameworks after the successful policy reform
The long, expensive, and complex road of clinical trials with vulnerable patient populations
The significant challenge of training a sufficient number of therapists
Ensuring broad insurance coverage
And many others
Ultimately, this ‘come down’ is a good thing.
It does not mean that the investment, commercial development, or political action should or will stop, but rather that sufficient time has passed, evidence has amounted, and discourse evolved to recognize that like any other technological, cultural, or political movement that gets ahead of itself, this too will require course correcting.
As you can see from the responses to Greg’s tweet, the surface area for such correction is significant.
So here we are, on the other side of the hype, facing a more complex and nuanced reality.
Clawing out of the trough of disillusionment means grappling with the hard questions and building the infrastructure and operational layer that will enable safe, responsible, and broad access to psychedelics.
With this in mind, on to the week’s featured developments.
🛁 DR. Bronner’s Covers KAP for Employees
Insurance reimbursement will be a primary means of creating access to psychedelic therapy in safe and responsible settings.
54% of Americans receive health insurance through their employer. Therefore, a significant challenge and opportunity will be employer-sponsored plans to cover these therapies, at least in the US.
In this direction, Dr. Bronner’s announced all employees would have coverage for Ketamine Assisted Therapy (KAP).
From a company press release:
“Dr. Bronner’s… has expanded its mental healthcare benefits to include Ketamine Assisted Therapy, as a first step in providing access to Psychedelic Assisted Therapy to employees to promote mental health. This innovative benefit plan is administered by Enthea, a non-profit healthcare organization responsible for medical policy development, provider network management, and benefit plan administration. Enthea establishes high ‘quality of care’ standards for the treatments offered, including credentialing and managing a network of specialty providers.”
Enthea is part of the Psychedelic Healthcare Infrastructure Projects, a group of organizations aiming to, as the name implies, create the infrastructure required for integrating PAT into healthcare systems.
As discussed above, these projects may not be sexy or headline-worthy in mainstream outlets but are necessary for safe, responsible, and broad access to psychedelic medicine.
An essential piece of the insurance reimbursement equation is the Cost-Effectiveness of PAT compared to other treatment options, which leads us to the next development this week.
💸 Cost-Effectiveness Analysis of MDMA Therapy
In order for payers to cover psychedelic therapies or any new treatments for that matter, they need to understand the cost-to-benefit analysis.
Ideally, a new treatment would yield better outcomes for a lower cost than currently available options. This appears to be the case for MDMA therapy compared to the current standard of care.
This week, researchers from the Global Initiative for Psychedelic Science Economics published Updated cost-effectiveness of MDMA-assisted therapy for the treatment of posttraumatic stress disorder in the United States: Findings from a phase 3 trial.
From the abstract:
"Compared to standard of care for 1,000 patients, MDMA-AT generates discounted net health care savings of $132.9 million over 30 years, accruing 4,856 QALYs, and averting 61.4 premature deaths. MDMA-AT breaks even on cost at 3.8 years while delivering 887 QALYs. A third MDMA session generates additional medical savings and health benefits compared with a two-session regimen."
When I asked how we should understand these results, lead author Elliot Marseille told me:
“It's not only cost-effective, but it's also cost-saving. It's not just good value for the money, but there's no net cost to it. You get tremendous health benefits and it saves the payer money. It is what we call a “dominating option” in my trade. Compared with the standard of care, it both provides more benefit and it costs less money.”
So, how do we ground this information in the larger context of pumping the breaks on the “psychedelics as miracle cure” narrative?
Admittedly, it is pretty hard.
But first, consider that this analysis is based on the cost of the trial ($11,537). I have seen estimates that a course of MAT therapy, upon approval, will cost anywhere from $15,000 to $50,000.
Second, proponents should be mindful that the perceived safety and effectiveness declines after FDA approval for nearly all drugs. Contributing factors for this include the fact that many patients who will ultimately receive the drug upon approval are excluded from research.
Finally, despite the potential as a dominating option, the upfront costs of MAT, the combination with psychotherapeutic support, and the yet to be determined drug labeling and Risk Evaluation and Mitigation Strategy (REMS) are constraints that warrant caution.
🔥 Expanding Access & Training: Fireside Equity Fund
From Techcrunch:
“Fireside recently launched a $200K fund that is available to its volunteers... After they’ve completed a year of volunteering with Fireside, they will be able to apply for up to $10,000 for initiatives that make psychedelic medicine more available to a broader group of people.”
Despite the promise as therapeutic tools, psychedelic experiences can be challenging, destabilizing, and occasionally harmful.
Fireside is attempting to create a virtuous cycle that on the one hand connects people undergoing difficult psychedelic experiences—by phone or mobile app— with trained volunteers who can provide “Real-time support - for when time doesn't seem real.”
Fireside hopes this volunteer opportunity can also serve as onboarding to a career in psychedelic therapy for underrepresented groups.
The recently announced Fireside Equity Fund is available for volunteers after completing a year of service on the line.
“After affinity volunteers complete a year of service, we’ll help those who wish to pursue further studies in the field of psychedelics and become Fireside Fellows through several pathways: The Fireside Equity Fund offers grants up to $10,000 to Fireside Fellows to pursue further education, training, and/or business development in the field of psychedelics.”
The lion's share of the “access” conversation revolves around the expected cost burden of future psychedelic treatments for patients, which are expected to be in the tens of thousands of dollars for a course of treatment.
However, the cost burden for therapists is another significant barrier to expanding access.
Upon completing a master’s or doctoral in psychology or counseling, would-be therapists will need to undergo specific training for psychedelic-assisted such as those offered by MAPS, Fluence, and others.
There is still significant uncertainty about how this all plays out. Still, one thing that can be addressed now is onboarding would-be psychedelic therapists with backgrounds that are not sufficiently represented.
In this way, Fireside can create a virtuous cycle to fill a significant need.
That’s it for this week. Thanks for reading!
Zach
This section has been edited to include a reference to Dr. Yaden’s use of the Hype Cycle several weeks before the writing of this post in presentations given at Imperial College London and Oxford. While I did not see his presentation I have a strong suspicion the idea to use the Gartner Hype Cycle Framework percolated through to me via conversations on Twitter from those who did.